Northern Red-Legged Frog – Photo by Corey Bunnell
Threats to a Species at Risk
The northern red-legged frog is a medium-sized frog in the family of “true” frogs (Ranidae). It is listed as a species of Special Concern under Canada’s Species At Risk Act (COSEWIC, 2004) and is assigned provincial Conservation Priority 1, highest of the 6 priority classes in British Columbia. In British Columbia the species is found in the southwestern part of the province, including Vancouver Island and the Gulf Islands. On the mainland, the species occurs west of the Coast Mountains in the Fraser Valley.
Here we summarize major findings of the first six years of study of the species in the Little Campbell River watershed. For details of methods and results see Bunnell et al. (2016).
Adjacent upland habitat: Wetlands are critical for breeding, but up to 90% of an adult red-legged frog’s life is spent in forested habitat (Maxcy 2004). Age of the forest does not appear to have a strong effect, although the species prefers abundant downed wood which is more common in older forests. Forest cover is also important to breeding areas. Size of wetlands had much less effect on egg masses produced than did surrounding vegetation. Egg masses increased significantly with the proportion of the wetland surrounded by forest and decreased significantly with the proportion of wetland surrounded by turf grass.

Values in Figure 1 are means for three classes of mean egg mass containing 11 sites each. Sites having 17 to 60 egg masses averaged only 11.9% turf grass, while sites with 0 to 2 egg masses averaged 44.9% turf grass. The most productive site (mean egg abundance of 60.3) had no turf grass in its 50-m buffer. The proportion of forest in a 50-m buffer around wetlands had a strong effect on red-legged frog egg mass abundance. Wetlands with less than 25% of the shore forested were significantly less productive than other sites. Egg masses increased until 96% of the buffer was forested. That in turn reveals that while narrow buffers around breeding sites may sustain breeding populations for a period, they are nowhere near as effective as wider buffers. We are aware of no similar analyses in the Pacific Northwest. Note: the cover data used in this analysis were from 2012; we intend to repeat the analysis with newly available GIS data.
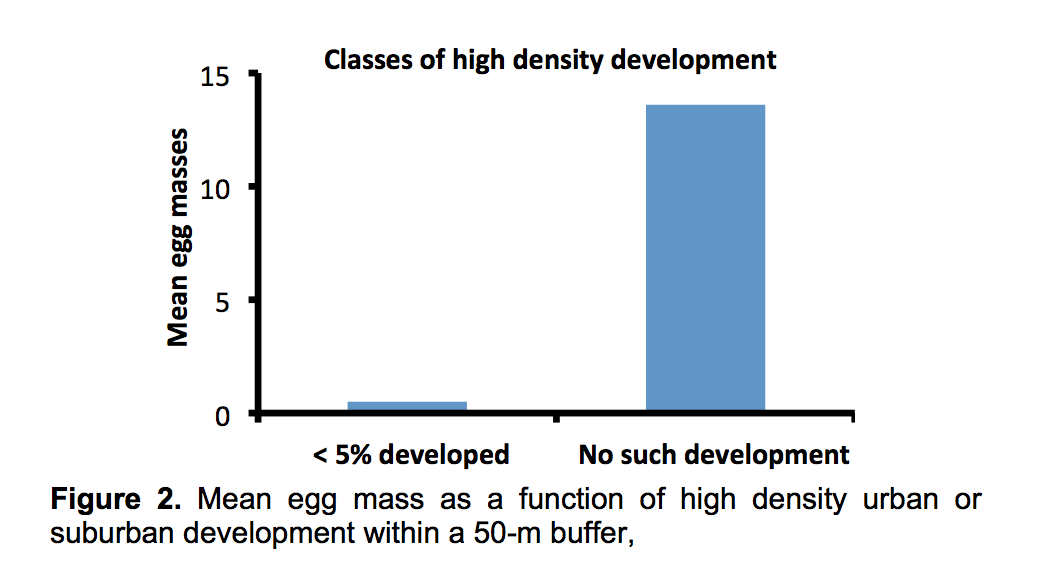
A third feature of terrestrial habitat also had strong effects on the abundance of egg masses – the proportion of high-density urban-suburban development.
Even when high density development within a 50-m buffer was less than 5%, mean egg mass was reduced by 96% compared to sites with no high-density development (from 13.6 to 0.5 mean egg mass). Low density development had much less effect. The most productive wetland (mean egg mass of 61.3) had 11.2% low density development.
Serious threats to the terrestrial habitat of red-legged frogs include:
- Reduction of forested habitat; narrow buffers around riparian or wetland areas are insufficient.
- Replacing natural vegetation with turf grass.
- Introduction of urban or suburban developments. These are particularly detrimental per unit area, likely because they bring associated hazards, including lower water quality.
Wetland habitat: is important for a relatively short portion of the frog’s life, but it is critically important then. Eggs take about 5 weeks to hatch and the hatchlings need another 3 to 4 months to metamorphose into frogs (Brown 1975). Several features of wetland habitat had negative effects on egg masses.
Egg masses declined significantly with various forms of water pollution indexed by increasing pH, decreasing dissolved oxygen and greater conductivity. Egg mass associated with extremes of these measures were consistently the lowest observed, averaging less than 1 egg mass per site.
Mean egg mass also declined significantly with increasing surface water temperature. Northern red-legged frog eggs are more susceptible to overheating than most other frog eggs so require relatively stable water temperatures (e.g., Frogwatch). That likely is one reason why wetlands bordered by forest and shade are more productive.
Northern red-legged frogs attach their egg masses to stalks of emergent vegetation (e.g., rushes and sedges) in quiet water of little or no flow (Storm 1960; Briggs 1987; Richter and Azous 1995). Some sites in the Little Campbell watershed host too little emergent vegetative to be productive. Average emergent class for sites with mean egg mass of <3 averaged 1.15, but were 2.63 for sites averaging more than 3 egg masses. Emergent class 1 is 0 to 10%; emergent class 3 is 25 to 50%.
The tall stature and robust leaves of non-native, invasive, reed canary grass (Phalaris arundinaceae) create a dense vegetative mat that suppresses other plant species and can be a barrier to small amphibians, like northern red-legged frogs. American bullfrogs are sufficiently robust to be unimpeded. Abundant Phalaris apparently constrains success of small frogs beyond limiting movement (Hayes 1994; Platt 2014). Rittenhouse (2011) found that the pulse of organic matter created by large stands can create anoxia. Bullfrogs cope better at lower levels of oxygen than do red-legged frogs. Currently, there is little evidence that Phalaris is depressing red-legged frog abundance in the watershed. Left unchecked, however, expanding Phalaris will encourage a decline in red-legged frogs.
Climate change and development will gradually reduce the water available to wetlands. Detrimental impacts on frog reproduction were not apparent in the 6 years of data reported here, but negative trends are well established. During summer the Little Campbell River already flows both ways, to and away from the ocean, as it dries out part way along. Unlike rivers that supply Vancouver’s drinking water, the Little Campbell is not replenished by deep snowfalls each year. Aquifers play a much larger role in the water flow of the Little Campbell. All water is connected. Currently, the sources of the Little Campbell are being depleted. Observation well #7 in Langley shows an average decline in aquifer water level of 0.225 m per year (see http://www.env.gov.bc.ca/soe/indicators/water/groundwater-levels.html), but the rate has been increasing since 1983. Actions to slow the decline are likely as drinking water declines.
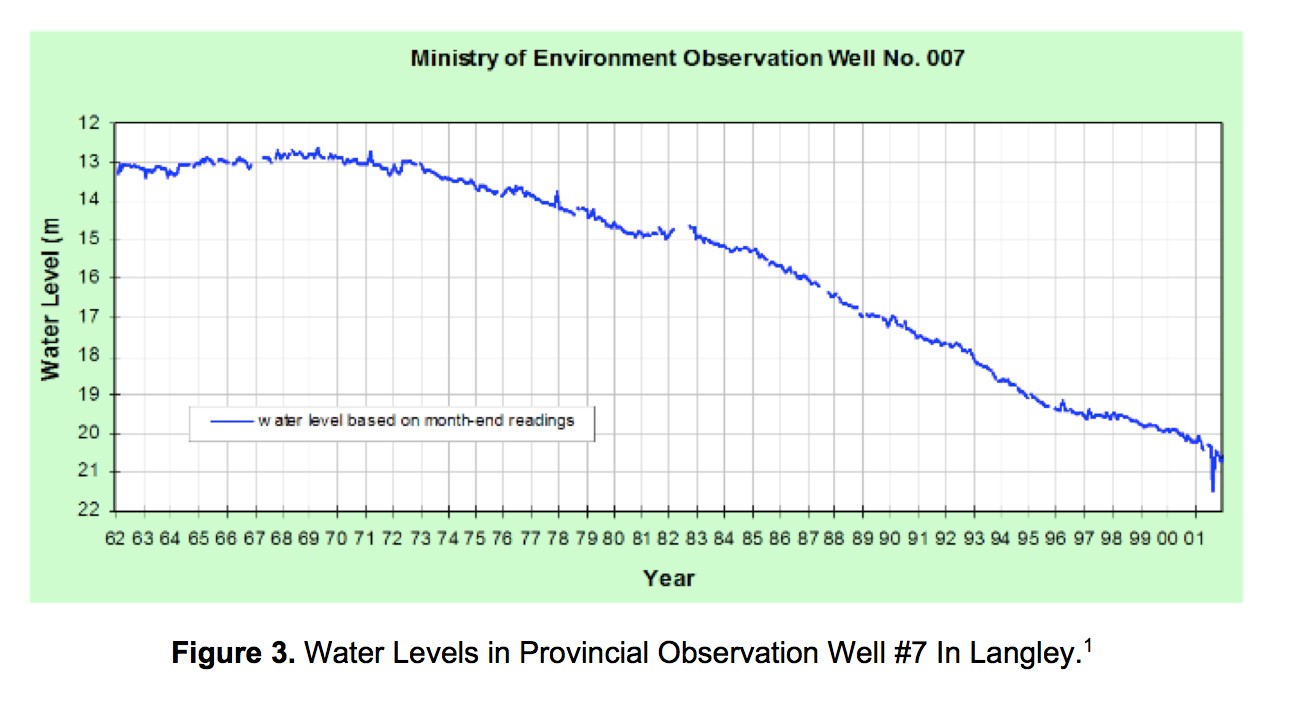
Other observation wells reflecting water levels in the Little Campbell River show similar patterns. Analysis of data indicates dropping water levels are not due to changes in precipitation but are the result of overuse of groundwater. A Rocha intends to monitor wetland depths more frequently in the continuing studies.
Serious threats to the wetland habitat of northern red-legged frogs include:
- Water pollution. Some wetlands already are seriously polluted.
- Warming water levels. This trend will worsen as the pattern in Figure 3 continues.
- Climate change will augment the trends of less water and warmer water.
Currently there is no established trend of reduction in emergent plants. Reed canary grass, however, spreads aggressively. Patient restoration can change that in limited, key areas. Experience at A Rocha Canada’s Brooksdale Environmental Centre indicates that cutting down Phalaris and planting red alder will eventually reduce Phalaris.
Introduced species present in the watershed that feed on northern red-legged frogs include pumpkinseed sunfish and bullfrogs. No efforts have been made to survey the pumpkinseed sunfish. They are a much smaller threat than the bullfrogs. Bullfrogs are an unwise and largely unintended introduction to the watershed. They are the largest frog in Canada and eat any animal that fits into their mouths. Red-legged frogs fit. Bullfrogs have completely or almost completely eliminated red-legged frogs from Stanley Park, areas of Delta and southern Vancouver Island.

Bullfrog in Campbell Valley Park – Photo by Anthea Farr
Bullfrogs were seen on 95.6% of the 90 visits to sites having 3 to 20 as peak egg mass values. Bullfrogs were seen much less often at sites having >20 as peak egg mass values (22.4% of 67 visits). Sites showing good egg production were less often exposed to bullfrogs. Sites having peak egg mass values of <3 had bullfrogs present on 32% of 93 visits, suggesting that these sites were favourable to neither species.
Compared to red-legged frogs, bullfrogs do better in warmer temperatures, lower levels of oxygen, higher levels of Phalaris and moderate levels of pollution. In short, current trends all favour bullfrogs over red-legged frogs. Without some changes in these trends red-legged frogs in the Little Campbell watershed appear destined to follow the demise of populations documented in Stanley Park, Delta and southern Vancouver Island.
Literature cited
Briggs, J.L.S. 1987. Breeding biology of the cascade frog, Rana cascadae, with comparisons to the R. aurora and R. pretiosa. Copeia 1987(1):241–245.
Brown, H.A. 1975. Reproduction and development of the Red-legged frog, Rana aurora, in Northwestern
Washington. Northwest Sci. 49(4):241–252.
Bunnell, C., F. Bunnell, A. Baylis, and C. Juteau. 2016. Long-term monitoring of northern red-legged frog (Rana aurora aurora) in the Little Campbell River – 2009-2014.
COSEWIC 2004. COSEWIC assessment and update status report on the Red-legged Frog Rana aurora in Canada. Committee on the Status of Endangered Wildlife in Canada. Ottawa. vi + 46 pp. (www.sararegistry.gc.ca/status/status_e.cfm
Maxcy, K.A. 2004. Red-legged Frog (Rana aurora aurora) Species Account in Forest Practices Code: Accounts and Measures for Managing Identified Wildlife. BC Ministry of Forests and BC Ministry of Environment, Victoria, BC.
Richter, K.O. and A.L. Azous. 1995. Amphibian occurrence and wetland characteristics in the Puget Sound Basin. Wetlands 15(3):305–312.
Storm, R.M. 1960. Notes on the breeding biology of the red-legged frog (Rana aurora aurora). Herpetologica 16:251–259.
Township of Langley. 2008. Township of Langley Water Management Plan. http://www.waterbucket.ca/okw/sites/wbcokw/documents/media/86.pdf